Published in: Synthesis
DOI: 10.1055/a-2755-9816
Authors: Priyadarshi Manna [TCGLS Member], Rusmita Mukherjee [TCGLS Member], Mrinalkanti Kundu[TCGLS Member], Susanta Sekhar Adhikari
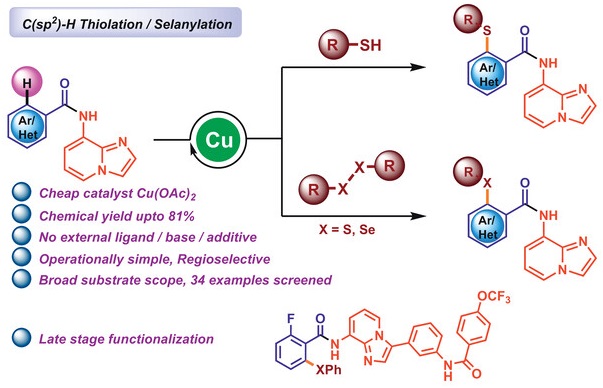
Abstract: Diaryl sulfides, diaryl selenides, and their derivatives are crucial scaffolds in the fields of organic chemistry and drug discovery. We report a practical and broadly applicable strategy for additive-free copper(II)-catalyzed ortho-sulfanylation and selanylation of β-C(sp2 )–H bonds in (hetero)arenes, directed by 8-aminoimidazo[1,2-a]pyridine (8-AIP), a removable 6,5-fused bicyclic N,N-bidentate directing group. This protocol features mild reaction conditions that enable the formation of diverse functionalized thioethers and selenides using thiols, disulfides, and diselenides, with enhanced functional group tolerance and unique site-selectivity. The present protocol is reproducible in gram scale, and, in addition, we have also the showed cleavage of chelating auxiliary