Published in: ACS Bio Med Chem Au 2025, 5, 5, 821–839
DOI: 10.1021/acsbiomedchemau.5c00058
Authors: Malkeet Kumar, Anees Ahmad,Anna Caroline Campos Aguiar,Sarah El Chamy Maluf,Anwar Shamim, Mariana Ferrer, Guilherme E. Souza, Marcos L. Gazarini, Dhelio B. Pereira, Thomas W. von Geldern, Delphine Baud, Barry Jones, Susanta Kumar Mondal [TCGLS Member], Paul A. Willis, Rafael Victorio Carvalho Guido, and Luiz Carlos Dias
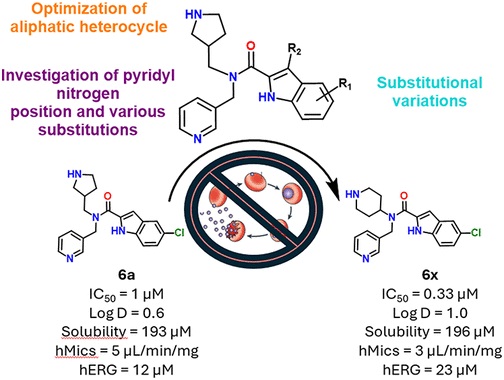
Abstract: Malaria still stands out as one of the most devastating and prevalent diseases globally, where the rise of resistance to different antimalarial drugs in different regions has posed significant obstacles to global treatment and elimination. Consequently, there is a pressing need for the development of new antimalarial agents with novel modes of action. In this study, we report the identification and optimization of new indole-2-carboxamide derivatives where structural modifications have yielded new compounds 6x with enhanced potency (Pf3D7-IC50 ∼ 0.3 μM) and improved metabolic stability (hMics = 3 μL/min/mg), while also minimizing the human ether-a-go-go-related gene (hERG, IC50 > 20 μM) channel activity and cytotoxic effect on hepatic cells (CC50 > 30 μM). Mode-of-action investigations revealed that a representative compound from this series interfered with homeostasis of the parasite’s digestive vacuole. However, cross-resistance was observed with resistant strains, which was linked to efflux pumps such as Plasmodium falciparum chloroquine resistance transporter (PfCRT). Despite this challenge, these indole-2-carboxamides provide versatile molecular templates for innovative medicinal chemistry to overcome cross-resistance while maintaining other attractive properties of this novel series.